Valence electrons are the electrons present in the outermost shell of an atom. That is, we will finally say that there are electrons equal to the atomic quantity within the iron atom. From the periodic table, we see that the atomic number of iron is 26.
Valence electrons are electrons which would possibly be found within the outermost ring of an atom. Because they are within the outermost ring, valence electrons play an necessary position in forming bonds with other atoms. In the sections ahead, we’ll uncover the significance of valence electrons and be taught extra about how precisely they play a job in creating new compounds. But for many of the transition and inside transition elements, the valence electrons are the electrons current within the shells exterior the noble gasoline core. Therefore, group quantity fourteen is also called the carbon group.
How Do Valence Electrons Relate To The Periodic Table?
Orange coloured parts are d-block parts and purple parts are the f-block elements. The components in blue color are the s-block parts. Valence electrons may also be decided because the electrons current in the shell with highest principal quantum quantity . • The durations of the periodic desk point out the number of shells that encompass a nucleus. Valence electrons are additionally known as outer-shell electrons. These compounds all have excessive melting factors and boiling points, as might be expected for ionic compounds.
- Whereas, valence electrons are related to elemental characters.
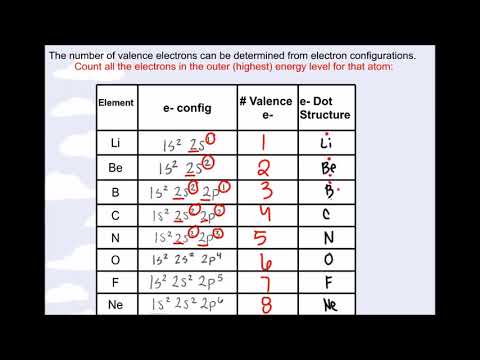
- Here is the list of periodic desk of parts with their valence electrons.
- Yes, my curious friends – this is chemical bonding.
- The shape of an s orbital is best described as a spherical form.
For brevity, many chemists record the electron configuration of an atom by giving solely its outermost subshell, like four s 1 for potassium or four s 2 for calcium. These electrons are most distant from the optimistic nucleus and, due to this fact, are most easily transferred between atoms in chemical reactions. However, there are occasional exceptions to the rule (e.g. for transition metals, a valence electron can additionally be within the inside shell, so it can have multiple type of valence). Nevertheless, at GCSE stage, it’s helpful to characterise elements by their valence outermost electrons and electron configuration. An atom’s electrons are grouped in distinct orbits.
TiCl4 and Mn2O7, then again, are each liquids at room temperature, with melting points under 0oC and comparatively low boiling factors, as may be anticipated for covalent compounds. They also dissolve in water to give aqueous solutions that conduct electricity, as could be anticipated. Valence electrons This is the presently selected merchandise.
Group-2 elements are called alkaline earth metals. The valence electrons of all the weather in this group are two. For instance how many valence electrons does nickel have, the electron configuration of magnesium exhibits that there are two electrons within the final orbit of magnesium.
Success In Chemistry
Both valency and valence electrons are applied for any chemical element. This is a color-coded desk made up of many different squares that lists the entire chemical elements known to humankind. You can often find these inside the duvet of chemistry textbooks. There is also a superb interactive table obtainable on-line right here. The periodic desk is split into four blocks by electron configuration.
Losing two electrons is tougher than losing one. As a result, they are less reactive, and these metals are more sturdy than group 1 elements. Four of the valence electrons are in lone pairs, which means that to be able to obtain an octet configuration, the oxygen atom must interact in two single bonds or one double bond. By locating the factor on the periodic table, the table is sectioned off into s,p,d, and f blocks. Whichever portion of the desk the component falls underneath will present its valence electron sub-shell. By determining which period the factor is located inside determines the power stage of the valence.
So to find a way to discover the valence electron you want to check the final digit of the series. I came across this website while i was engaged on my science project. They have some distinctive and interesting options like comparability of metals, Metals Quiz, plot graphs, examine statistics and far more! Core electrons experience the full charge of the nucleus.
The whole variety of electrons within the last shell after arranging the electrons of the element is known as the valence electron. Depending on the element, the valence electrons may be paired or unpaired. When solely seven electrons are present within the outermost shell of an atom, then the atom is said to own seven valence electrons. Elements having seven outer-shell electrons are current in group (17) of the Periodic Table.
Definitions For Calculating Valence Electrons In A Molecule Or Polyatomic Ion
For example, the electron configuration of sodium reveals that sodium is an element of group-1 and that sodium has one electron in its last orbit. The place of an atom within the periodic desk represents the number of valence electrons present within the outermost shell of the atom. Across each row or interval of the periodic table, the number of outer-shell electrons in teams (1 – 2) and (13 – 18) will increase by one from one component to the next.

Najnowsze komentarze